What is SMA?
Neuromuscular disease with multi-systemic aspects
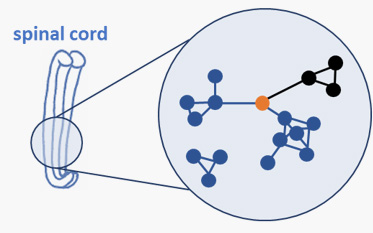
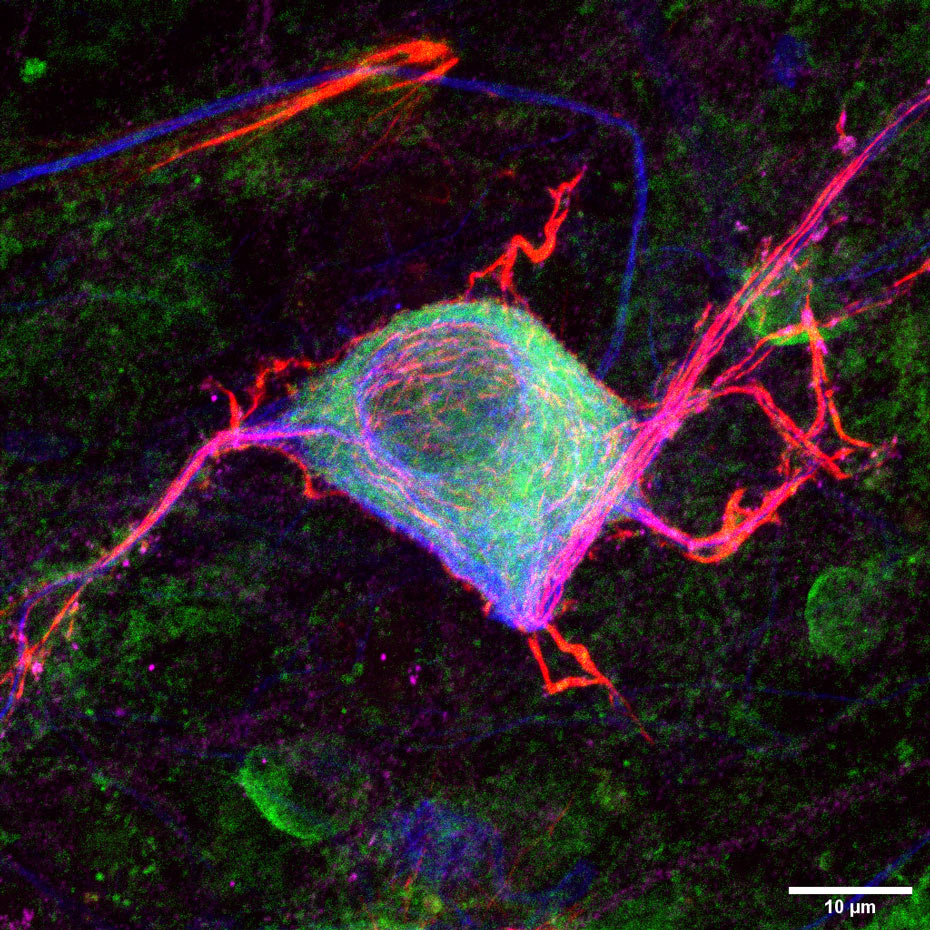
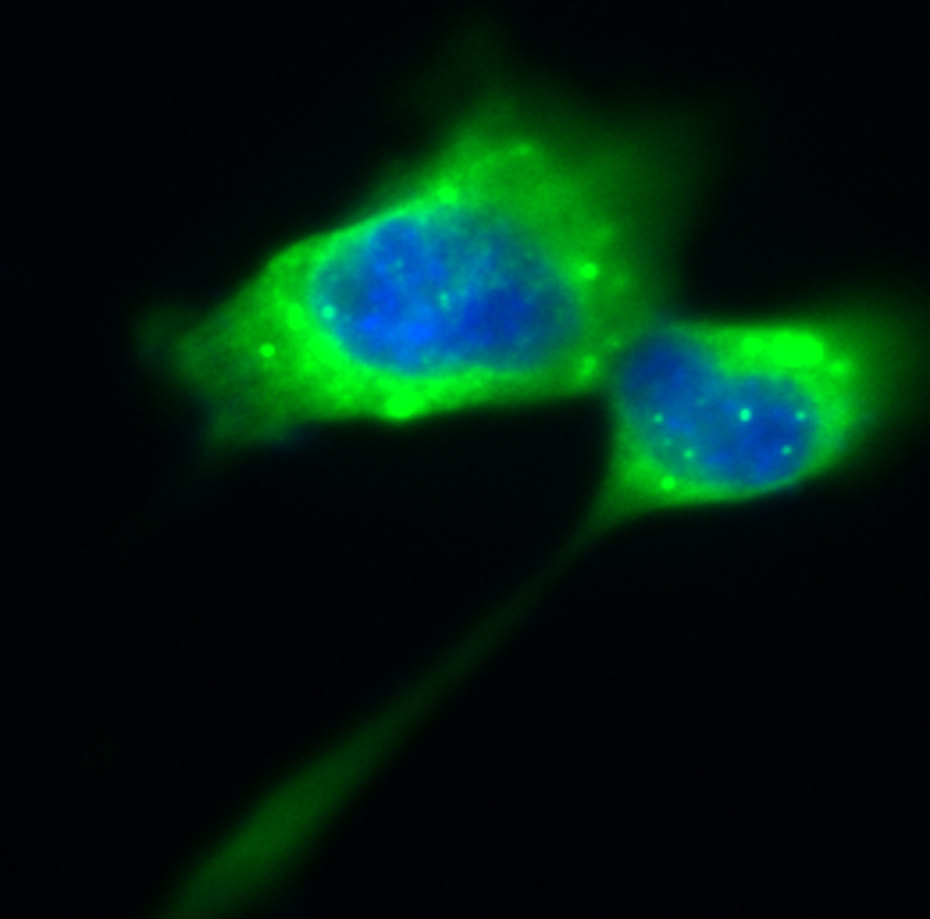
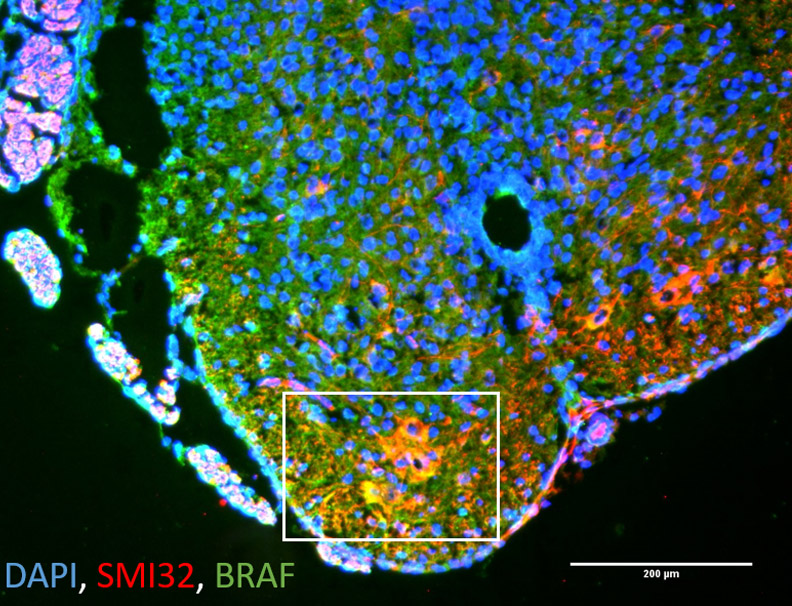
Spinal Muscular Atrophy (SMA) is a monogenic neurodegenerative disease affecting motoneurons in the spinal cord and brain stem. Mutations of the Survival of Motoneuron (SMN) gene 1 lead to neurodevelopmental defects and degeneration of the neurons responsible for innvervation of muscles. Due to the lack of the SMN protein in all organs and tissues, SMA is considered now to be a multi-systems disorder.
SMABEYOND
Besides motoneurons in the central nervous system (CNS), there is growing evidence of an involvement of peripheral organs in SMA. The project SMABEYOND addresses those functions on molecular, cellular and systems levels both in SMA models and patients.
SMABEYOND is an international consortium of laboratories from Spain, United Kingdom, The Netherlands, Italy and Germany and is funded by the European Commission. One aim of the project is to investigate the role of the SMN protein in peripheral organs and determine the intrinsic defects encountered in SMA patients. The ultimate goal is to develop SMA patient-derived disease models and test the therapeutic effect on peripheral organs of systemic SMA therapy (MSCA-ITN-ETN - European Training Network, grant agreement ID: 956185).
Neurodegenerative Diseases
SMA is a neurodegenerative disease among others also affecting motoneurons, but also other types of neurons in the nervous system. We use our expertise to develop novel treatment approaches for specific other diseases.
Neuronal
Infections
SMATHERIA uses its expertise about dysregulated molecular networks in neurons to unravel mechanisms of viral infections of the nervous system.
Treatments and Therapy
We aim to improve current therapies by novel combinatorial approaches and develop strategies for the treatment of other diseases.